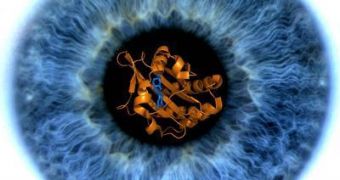
The main enzyme responsible for converting light to flows of energy for the brain has been detected for the first time, using high-resolution X-ray crystallography and nuclear magnetic resonance imaging. UW biochemist Clemens Heikaus, and Rutgers research associate Sergio E. Martinez are responsible for this discovery, which can lead to a better understanding of the way light stimulates cells inside the retina to produce electrical impulses which are then transformed into vision sensations inside the brain.
Human retina has two types of photo receptors, ones that look rod-like and other ones shaped like cones. The rod receptors are responsible for night vision and motion sensation, while the conic ones deal with light regulation inside the eye, color sensitivity, daylight vision and acuity. While both contain the phosphodiesterase 6 (PDE6) enzyme, the role of this particular substance in cone receptors has been largely un-researched.
It seems that a portion of the complex PDE6 enzyme, dubbed GAF A Domain, binds to a specific tiny messenger molecule known as cGMP, which regulates the activity of the enzyme. According to researchers, the connection is super-sensitive, meaning that the GAF A Domain always binds with the same molecule very quickly. This allows for high-speed information captured by the enzyme to be transmitted to the brain almost instantaneously, giving humans the impression they can see everything that's happening around them in real time. In fact, all human receptors throughout the body need a very short amount of time to send all data to the brain.
Having discovered that the enzyme only activates when the domain binds with the messenger molecule, scientists now think they've precisely located the "on-off" switch for electrical impulses for the brain. They hope to someday use this discovery to treat severe retina diseases, such as night blindness and retinitis pigmentosa, which affect tens of thousands worldwide.
 14 DAY TRIAL //
14 DAY TRIAL //